“For many years I was taking opiates to deal with the pain from both injuries and surgery. An alternative method of pain control will be a welcome relief.”
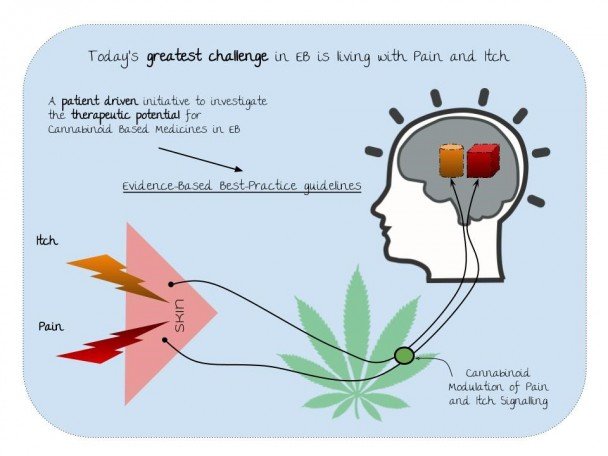
The charity DEBRA is funding a two-year project exploring cannabinoid medicines to treat adults with Epidermolysis Bullosa (EB).
Nicholas Schrader and Professor Marcel Jonkman will be heading the trails. Fifteen participants aged 18+ will take part in the study at the Centre for Blistering Diseases at the University Medical Centre Groningen in the Netherlands.
Therefore, in researching this potential treatment, this project aims to gain an insight as to whether it can ultimately improve the quality of life in patients with EB.
Globally, researchers are currently looking into various CBM formulations and administration forms for numerous diseases. In the Netherlands recent advances have led to the standardisation of cannabis plants for therapeutic use by Dutch patients. With regard to the CBM choice for this study, these “phytocannabinoids” (plant-based cannabinoids) are extracted from the plants and incorporated into an oil which is administered as droplets under the tongue (sublingually). The final product is a pharmaceutical – grade CBM oil, and is currently distributed to patients in the Netherlands who have obtained a prescription from their registered physician.
The use of plant-based CBMs is not physically addictive, and has not been associated with life threatening or debilitating complications. As there is currently not enough information about the long term effects of cannabinoid use on the developing nervous system in children, this study is only for patients over 18 years old. Specific inclusion criteria will take into account pre-existing psychiatric conditions as these can be affected by CBM use.
The research will investigate if additional treatment with this sublingual oil can improve the quality of life of adults with EB suffering from pain and/or itch. The effects of the CBM oil can last 4-8 hours and will therefore need to be taken 4 times daily to maintain blood levels. Patients will report pain, itch and changes in their quality of life monthly over 6 months. Quality of life, pain and itch will be measured by the use of patient questionnaires or patient reported outcome measurements. A number of these will be used to also identify which measurements are most sensitive to changes in patients with EB.
Stopping current medication is not required as a study participant, during the course of the research, the research team will monitor the change in medication-use of each patient (such as a reduction in opiate use), and at the end scrutinize whether this correlates with CBM oil use.
This study is considered a prospective, open label, proof of concept study – testing a new medicine in a small number of patients, where everyone takes the new medicine to see what benefits can be identified. Therefore, in researching this potential treatment, this project aims to gain an insight as to whether it can ultimately improve the quality of life in patients with EB.
Patients with EB face constant, debilitating symptoms such as pain and itching. Some patients have suggested that cannabinoid based medicines are effective for symptom control.
Caroline Collins, director of research at DEBRA, said: “We are extremely pleased to fund this clinical trial, particularly in light of the government’s decision to legalise some forms of medical cannabis.“The clinical trial will start to gather the scientific evidence needed to prove that CBMs are an effective treatment for pain and itch caused by EB and will begin the work towards a new treatment protocol and evidence-based guidelines for the management of these debilitating symptoms, which we hope will improve the quality of life for many.”
Simon Weston CBE, president of DEBRA, added: “For many years I was taking opiates to deal with the pain from both injuries and surgery. An alternative method of pain control will be a welcome relief.”
Source & Image DEBRA.org